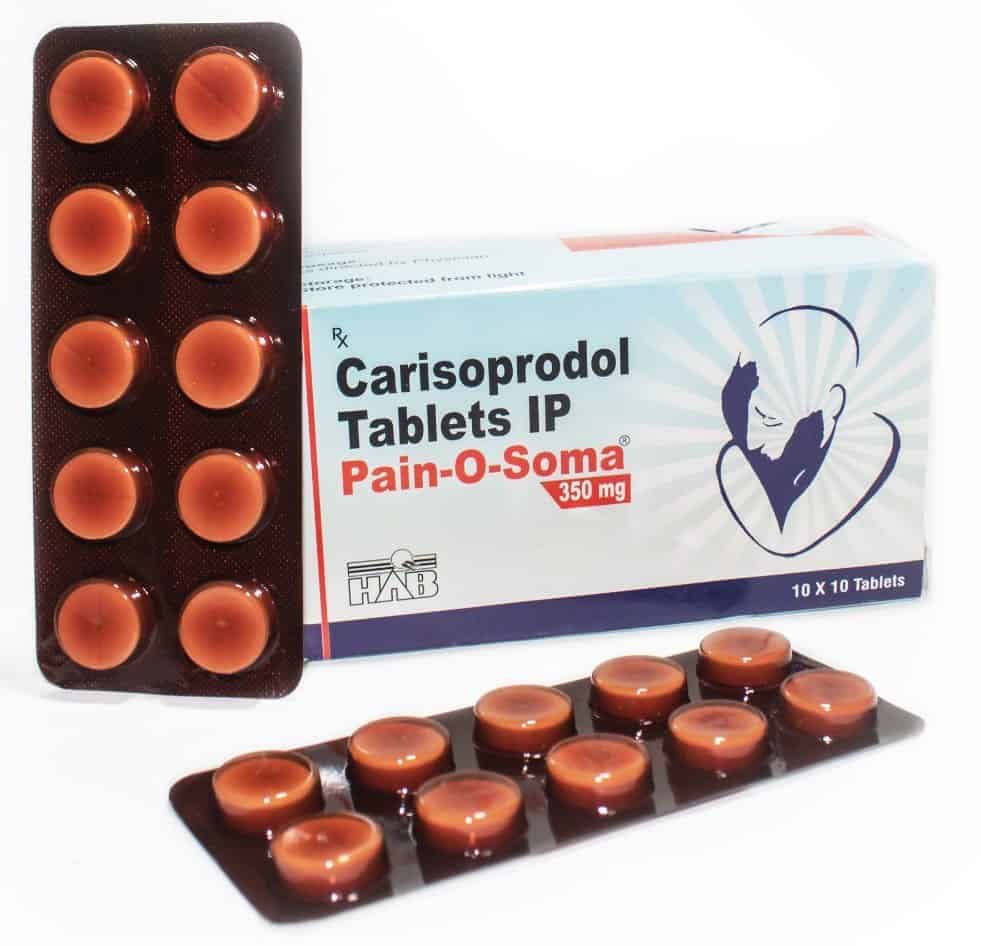
Carisoprodol
$46.75 – $119.00Price range: $46.75 through $119.00
Shipping: 24h Dispatch with Fast ‘N Insured – US Shipping | See details
Guarantee: Money Back Guarantee with Free Tracking | See details
Quality: Independent Product Analysis | View certificate
Payments: Buyer Protected via ![]() | See details
| See details
Carisoprodol is a short-term treatment for muscle pain and discomfort, often used alongside rest, physical therapy, and other interventions. Its mechanism of action involves muscle relaxation.
Prescription and Utilization:
Soma, a brand name for the generic drug Carisoprodol, is a pharmaceutical agent prescribed by medical professionals for the management of muscle pain and associated discomfort. Its widespread usage is attributable to its notable sedative properties.
Potential for Habituation:
However, it is essential to acknowledge that Soma’s sedative effects have the potential to lead to habituation in individuals. This habit-forming tendency may prompt unsanctioned usage by certain segments of the population.
Recreational Use and Abuse:
Beyond its legitimate medical use, Carisoprodol is subject to recreational misuse across various age groups. Individuals may misuse the drug to attain a euphoric state, thereby inducing a cycle of addiction and dependence.
Primary Medical Utility:
Despite these issues, it is pertinent to recognize that Carisoprodol primarily functions as a muscle relaxant, making it a valuable recommendation by healthcare providers for alleviating musculoskeletal discomfort. This dual nature of the drug underscores the importance of prudent medical application while addressing its potential for misuse in the broader context of public health.
Dosage:
The recommended dosage of Carisoprodol varies depending on individual patient factors and the severity of the condition being treated. Typically, the initial dosage for adults is 250 to 350 milligrams three times a day, and it can be adjusted as needed. The maximum recommended duration of use is up to two to three weeks, as prolonged use may result in tolerance or dependency. Patients are advised to follow their healthcare provider’s specific dosing instructions.
Usage Instructions:
Carisoprodol is administered orally, with or without food, under the guidance of a healthcare professional. It is imperative not to exceed the prescribed dose or duration, as doing so will not expedite therapeutic benefits and could heighten the risk of adverse effects.
Dosage Information:
The appropriate dosage of Carisoprodol is contingent on individual medical conditions and responses to treatment. Prolonged usage is not recommended, with a typical duration of up to three weeks, unless otherwise directed by a healthcare provider. Abrupt discontinuation of Carisoprodol may lead to withdrawal symptoms. In such cases, gradual dose reduction may be advised to mitigate withdrawal risk, particularly for long-term or high-dose usage. Patients with a history of substance use disorders should adhere strictly to prescribed dosages to minimize addiction risk.
Monitoring and Duration:
Should the condition persist or worsen after 2 to 3 weeks, consultation with a medical professional is advisable.
Common Adverse Effects:
Common side effects encompass dizziness, drowsiness, and headaches. Patients experiencing persistent or aggravated side effects are advised to promptly consult with a healthcare provider or pharmacist.
Serious Adverse Effects:
The medication may infrequently lead to addiction, especially in individuals with substance use disorders. It is paramount to strictly follow the prescribed regimen to mitigate this risk. Patients are encouraged to consult with their healthcare provider or pharmacist for additional information.
Reporting Serious Side Effects:
Prompt medical consultation is necessary if serious side effects such as confusion occur. Although rare, a severe allergic reaction to Carisoprodol may manifest as symptoms like rash, swelling, dizziness, or breathing difficulties. Any unlisted side effects should also be reported to a healthcare professional.
Warnings & Precautions:
Patient Allergies:
Before commencing treatment, individuals should disclose allergies to Carisoprodol or related substances to their healthcare provider, pharmacist, and inquire about potential inactive ingredient-related sensitivities.
Medical History and Conditions:
Prior to usage, patients should inform their healthcare provider or pharmacist of their medical history, particularly if they have conditions such as acute intermittent porphyria, kidney disease, liver disease, seizures, or a personal or family history of substance use disorders.
Caution with Drowsiness-Inducing Substances:
Carisoprodol may induce drowsiness, which can be exacerbated by alcohol or marijuana use. Engaging in activities that require alertness is not recommended. If using marijuana, consultation with a healthcare provider is advised.
Pre-Surgery Notification:
Prior to surgery, patients should inform their healthcare provider or dentist of all medications they are using, including prescription and non-prescription drugs, and herbal products.
Special Considerations for Older Adults:
Older individuals may be more sensitive to certain side effects, such as drowsiness and confusion, which could increase the risk of falls.
Pregnancy and Breastfeeding:
Pregnant individuals should use Carisoprodol only when absolutely necessary, following a thorough discussion of potential risks and benefits with their healthcare provider. The medication passes into breast milk and may have undesirable effects on nursing infants, necessitating a consultation with a healthcare provider before breastfeeding.
Storage and Disposal:
Carisoprodol should be stored at room temperature, shielded from light and moisture, and kept out of the bathroom. It should also be kept away from children and pets. Proper disposal in accordance with expiration or cessation of need should be facilitated through consultation with a pharmacist or local waste disposal company.
Drug Interactions:
Carisoprodol may interact with other drugs, which can alter their effectiveness or increase the risk of serious side effects. Patients should maintain a comprehensive record of all medications used, both prescription and non-prescription, as well as herbal products, and communicate these with their healthcare provider and pharmacist. Changes in medication regimens should not be undertaken without professional approval.
Overdose:
In case of an overdose with severe symptoms like loss of consciousness or breathing difficulties, immediate medical attention should be sought through calling 911 or contacting a poison control center. Overdose manifestations may comprise severe drowsiness, seizures, respiratory depression, altered mental state, motor impairment, unsteady movements, or visual disturbances.
Additional Information:
Missed Doses:
If a dose is missed, it should be taken when remembered. If the time for the next dose is near, skipping the missed dose is advisable. Doubling the dose is not recommended.
Restrictions:
Sharing this medication with others is illegal. Carisoprodol should only be used for the condition for which it was prescribed by a healthcare professional. Usage for alternative conditions should be directed by a healthcare provider, who may consider a different treatment.
Additional information
| Quantity of Tablets |
30 ,60 ,90 ,120 ,180 ,300 |
|---|